
Thomson set up his cathode ray tube with but placed a magnet above the path of the rays. He knew that electrically charged objects can be deflected by magnets (Michael Faraday discovered this and is his theory of electromagnetism). What are observations of cathode ray tube experiment What are the important conclusions drawn from above observations? AND if they already knew that the electron was small and negative then the atom must have a small positive nucleus with the electrons around them. If the positive alpha particles mostly passed through the foil but some bounced back. Rutherford’s experiment prompted a change in the atomic model. How did Rutherford’s experimental evidence led to the development of a new atomic model? The charge of an electron was measured by R. Thomson in Cathode Ray Tube (CRT) experiment. When elements react their atoms may combine in more than one whole-number ratio.Įlectron was discovered by J.
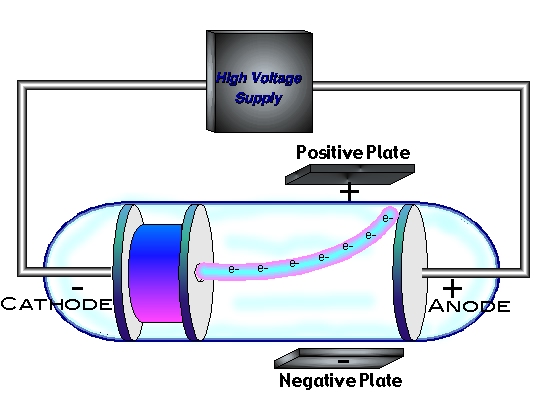
How did experimental evidence support the development of atomic theory?Įxperiments that Dalton and others performed indicated that chemical reactions proceed according to atom to atom ratios which were precise and well-defined. Is cathode ray an electromagnetic wave?Ĭathode rays are not electromagnetic wave because they do not have electric and magnetic components perpendicular to each other. The beam of electrons produced forms a cathode ray. Low pressure is maintained in the cathode ray tube so that there is enough space between the gas molecules so that ions formed in the tube can accelerate at high speeds thereby ejecting electrons and forming positive ions when they strike gas molecules. Why are cathode rays produced at low pressure? As they are energetic electrons when they strike a certain substance or the glass wall of the discharge tube this excites the atoms of the substance or the glass and cause them to emit light a glow called fluorescence. Why do cathode rays produce fluorescence?Ĭathode rays produce fluorescence in some materials. In addition he also studied positively charged particles in neon gas. He demonstrated that cathode rays were negatively charged. Thomson discovered the electron by experimenting with a Crookes or cathode ray tube. How did Thomson’s experiments prove the existence of electrons? This was probably one of the most significant experiments ever carried out. That he could also determine the charge of the electron was a secondary benefit. … This was the big result of the oil drop experiment. Millikan’s oil drop experiment proved that electric charge is quantized. The resulting radiation showed high penetration through a lead shield which could not be explained via the particles known at that time. In 1932 the physicist James Chadwick conducted an experiment in which he bombarded Beryllium with alpha particles from the natural radioactive decay of Polonium.

Rutherford also predicted the existence of neutral nuclear particles called neutrons but he failed to find them. He predicted-and soon discovered-that the nucleus contains positively charged particles which he named protons. Rutherford called the area of concentrated positive charge the nucleus. What did Rutherford think was going to happen? Dalton thought that atoms were indivisible particles and Thomson’s discovery of the electron proved the existence of subatomic particles. What two pieces of knowledge did Thomson learn from his cathode ray tube experiment about atomic structure?Įxplanation: Thomson’s experiments with cathode ray tubes helped him to discover the electron (which Dalton did not know about). These negatively charged particles are deflected from their straight-line path when an electric field is applied. … Thomson discovered that cathode ray particles carry a negative charge. Thomson discovered that cathode ray particles carry a negative charge. What did JJ Thomson’s cathode ray experiments determine quizlet? There are subatomic size particles in an atom. Matter contains negative charge and by inference positive charge.In the atom the particles of the cathode rays are embedded in a diffuse cloud of positive charge. Particles of the cathode rays are fundamental to all matter. Are cathode rays fundamental to all matter?Ĭathode rays have mass. … Protons and neutrons are found in the nucleus of an atom. How did cathode ray tube experiment lead to the conclusion that atoms contain electrons? because Thomson saw the ray move from the cathode to the anode so the particles have negatively charge. How did the cathode ray tube experiment lead to the conclusion that atoms contain electrons?
